Scientists have long tried to crack their secrets, which could lead to the Holy Grail of medicine: regenerating aging, diseased or damaged body parts.
It hasn’t been easy. Stem cells are delicate things that need pitch-perfect conditions to remain “blank” and keep dividing. Even the slightest disturbance can set them off on an irreversible journey to becoming, say, an eye. Worse, vibrations or changes in their environment can easily kill them.
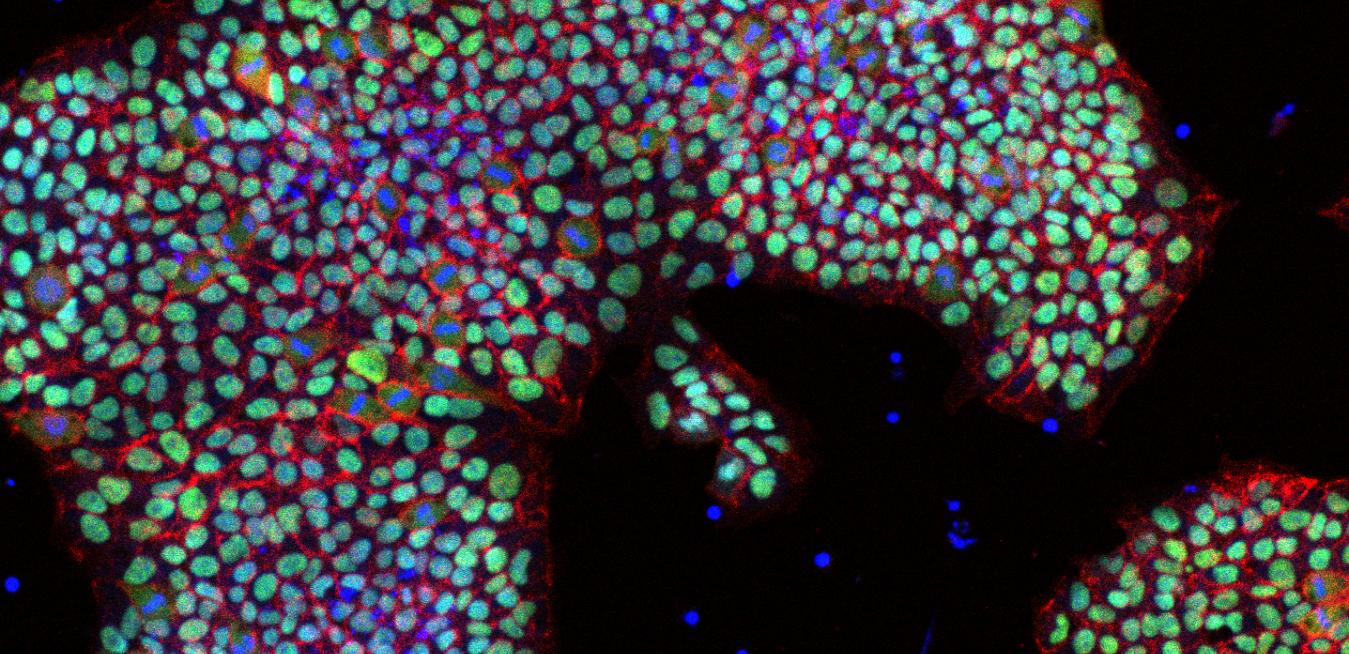
 Above: Pluripotent stem cells start out in the embryo as biological blank sheets but transform in developing animals and grow into tissues that become the brain, bones, heart, liver and the rest of our bodies. Image credit: Getty images. Top: An image of human pluripotent stan cells in IαI media. Image credit: GE Healthcare Life Sciences
Above: Pluripotent stem cells start out in the embryo as biological blank sheets but transform in developing animals and grow into tissues that become the brain, bones, heart, liver and the rest of our bodies. Image credit: Getty images. Top: An image of human pluripotent stan cells in IαI media. Image credit: GE Healthcare Life SciencesLike a school of sardines, they also clump together. If they lose contact with one another or even the bottom of a petri dish, they’ll die unless pre-treated with a protein that inhibits cell death. As a result, researchers have been forced to construct delicate protective and nourishing matrices by coating their Petri dishes with cocktails of proteins, polymers or feeder cells.
But simple times might be beckoning. Scientists from Sweden’s Uppsala University, the U.K.’s University of Nottingham and GE Healthcare Life Sciences say they’ve unlocked the secret to keeping stem cells happy with much less mess and fuss. If it takes off, it could scale up stem cell R&D and unleash new biotechnology market applications.
“What we’ve shown is the next step in producing stem cells at scale for research, pharmacological testing and to eventually make cells to replace diseased organs,” says Cecilia Annerén, global product marketing manager at GE Healthcare and a researcher at Uppsala University, who is leading the group that made the discovery. “This is the first time that you don’t need to pre-coat the dish to grow stem cells.”
Annerén’s team discovered that a protein found in blood serum, called inter-α-inhibitor (IαI), can help stem cells attach, survive and stay undifferentiated just by adding it in solution to a standard xeno-free growth medium, no pre-coating needed.
By mixing these ingredients together with the stem cells, researchers can pour the solution into dishes and the cells will attach to naked plastic, a big time-saver in this type of work. The advance can shave a day off of lab time that is normally consumed preparing media and dishes to harbor the cells. What’s more, IαI seems to support survival, and cells thrive in the mix while remaining undifferentiated. “It seems that the cells really, really like this protein,” Annerén says. “We see better survival after single-cell splitting and the cells grow more robustly, which simplifies the process and saves a significant amount of time.”
This use of IαI is new for science. The protein is produced in the liver and found in high concentrations in blood plasma. It forms complexes with the extracellular matrix and is known to be important in the process of inflammation.
Still, obstacles remain for IαI’s widespread adoption, Annerén says. Purifying it from the rest of the serum, which itself is expensive, is required. Thus, the next task is to figure out how to make the IαI protein cheaply and plentifully, so that the discovery could help ramp up stem cell production. Then, researchers would have all of the magical cells they need to study life’s development and unlock new ways to treat injury and disease. “This is a jump for the field,” she says. “It’s a big deal.”
Photo credit: Sara Pijuan Galitó | Uppsala University