A new AI could help people with paralysis write with their thoughts. A cancer therapy tricks tumor cells into destroying themselves. A nanoscale mechanical gear could be the hardest-working artificial molecule. This week’s coolest things are turning science and medicine inside out.

What is it? Stanford researchers found a way to “quickly convert [a person’s] thoughts about handwriting into text on a computer screen.”
Why does it matter? The technology, which involves an AI program and a brain-computer interface (BCI), could help people with paralysis or who can’t move their hands communicate with handwriting. “We’ve learned that complicated intended motions involving changing speeds and curved trajectories, like handwriting, can be interpreted more easily and more rapidly by the artificial-intelligence algorithms we’re using,” said research scientist Frank Willett, lead author of the study’s findings, which were published in the journal Nature. “Alphabetical letters are different from one another, so they’re easier to tell apart.”
How does it work? The study’s participant, a man researchers refer to as T5, was fitted with two BCI chips. The chips contained electrodes that picked up signals from the brain’s motor cortex, which controls hand movements. When T5 focused his thoughts on writing letters of the alphabet on paper with a pen, the BCI sent these signals to a computer, where the newly developed algorithm transcribed them into text on a screen. T5 repeated each letter ten times to teach the program his handwriting style. Ultimately, he set new speed records for copying sentences (around 18 words per minute) and “freestyle writing” (15 words per minute, triple the previous record from a keyboard-and-mouse setup).
What is it? Scientists at the University of Zurich have developed a cancer therapy that tricks tumor cells into destroying themselves without damaging healthy cells.
Why does it matter? “The therapeutic agents, such as therapeutic antibodies or signaling substances, mostly stay at the place in the body where they’re needed instead of spreading throughout the bloodstream where they can damage healthy organs and tissues,” says research group leader Andreas Plückthun. The Zurich team outlined the technology, called SHREAD (for SHielded, REtargeted ADenovirus), in the journal PNAS.
How does it work? SHREAD modifies a common respiratory virus called adenovirus and uses it as a Trojan horse to invade breast cancer cells and deliver genes that produce trastuzumab, an anti-cancer therapeutic approved to treat breast tumors. Within a few days, the tumors contained more of the antibody they produced on their own with SHREAD technology than when trastuzumab was injected directly. “We trick the tumor into eliminating itself through the production of anti-cancer agents by its own cells,” says Sheena Smith, a postdoctoral fellow who led the development of the delivery process. Concentrations in the blood and other tissues, which could cause side effects, were much lower. Now the team is working on applications for the COVID-19 vaccine.
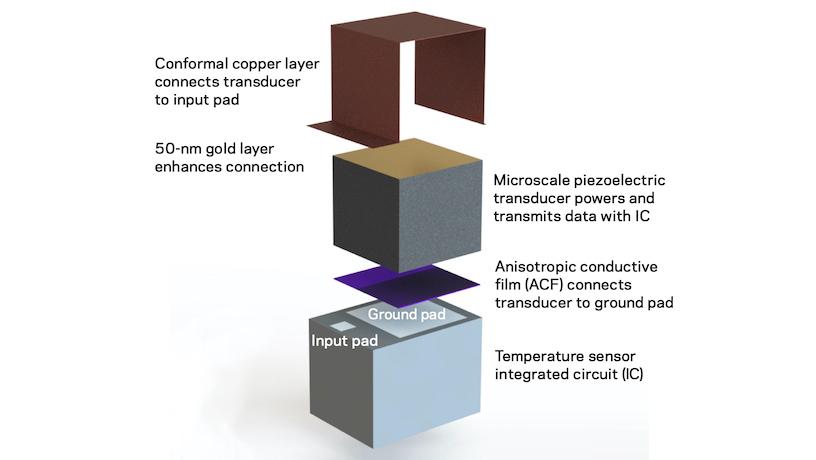
What is it? Engineers at Columbia University built the smallest-ever full electric circuit on a single chip, tiny enough to be implanted by a hypodermic needle and designed to monitor patients’ body temperature.
Why does it matter? Implantable medical devices have the potential to transform healthcare and everyday wellness. But they remain “highly volume-inefficient,” Columbia Engineering says, with multiple chips, wiring, external components, packaging and batteries all competing for space. “This is a chip that alone, with nothing else, is a complete, functioning electronic system,” says professor Ken Shepard, leader of a new study on injectable micro devices published in the journal Science Advances. “This should be revolutionary for developing wireless, miniaturized implantable medical devices that can sense different things, be used in clinical applications and eventually approved for human use.”
How does it work? To get down to the micro scale, chip designer Chen Shi, lead author of the published study, had to leave radio-frequency signals behind. Instead, the team used ultrasound, both to power the device and to communicate with it wirelessly. Because ultrasound waves are much shorter at a given frequency, this change allowed the engineers to pack a complete single-chip system with an attached antenna into one-tenth of one cubic millimeter. They also added piezoelectric materials, which generate electric charge in response to mechanical stress, directly to the circuit to act as a transducer. “We wanted to see how far we could push the limits on how small a functioning chip we could make,” Shepard says.
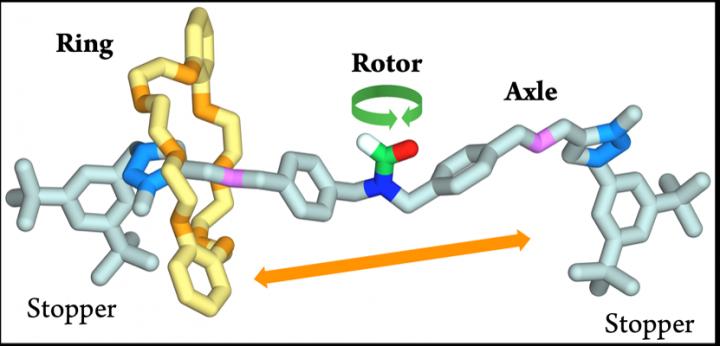
What is it? A team of scientists in Bologna, Italy, created NanoGear, a microscopic structure, complete with a rotor and axle, that could one day turn a molecular motor to power a nanoscale mechanical device.
Why does it matter? “The construction of molecular devices such as NanoGear is a first step forward towards the development of ultra-miniaturized mechanical devices based on molecular motors, with potential breakthrough applications in various fields of technology and medicine,” says Alberto Credi, co-leader of the team that developed NanoGear. A paper on the technology was published in the journal Chem.
How does it work? NanoGear includes a rotor, axle and ring. The rotor is chemically bonded to the axle, while the ring moves freely along it, mechanically “locked” by a wide stopper at each end. Since there’s no nano motor (yet), the rotor and ring move according to the molecule’s thermal energy, essentially rolling in neutral. At 65 degrees Celsius, the rotor completes about 260 rotations per minute, while the ring shuttles end to end about seven times. The team designed two blades on the rotor to increase visibility and used sophisticated nuclear imaging to observe the NanoGear in action.

What is it? Researchers created a water-based jelly that is stronger and more flexible than other hydrogels and could be used for 3D printing. It may have benefits for medicine and soft robotics.
Why does it matter? Water-based materials called hydrogels are gaining popularity for flexible 3D printing. A research team from North Carolina State University has created a “homocomposite” hydrogel by combining two different-sized networks of the same material, resulting in a more durable material that could form strong biological scaffolds or soft robotic materials.
How does it work? Scientists started with an alginate, a chemical compound found in seaweed and algae that’s already used medically for wound care. They created a micro-sized hydrogel from alginate molecules, then a second, nano-sized hydrogel from smaller particles of the same alginate. “Merged together they produce a jelly-like material that is better than the sum of its parts, and whose properties can be tuned precisely for shaping through a 3D printer for on-demand manufacturing,” says professor Orlin Velev, corresponding author of a study on the new material published in Nature Communications. Co-author Lilian Hsiao adds: “We are reinforcing a hydrogel material with the same material, which is remarkable because it uses just one material to improve the overall mechanical properties.”