Researchers used a gene therapy injection to restore sight to patients with a degenerative nerve disease, new batteries from Toyota could be a “game changer” for electric vehicles, and scientists came up with a new way to grow livers in the lab. Also in this week’s coolest scientific findings? Synthetic microparticles that are surprisingly good swimmers.
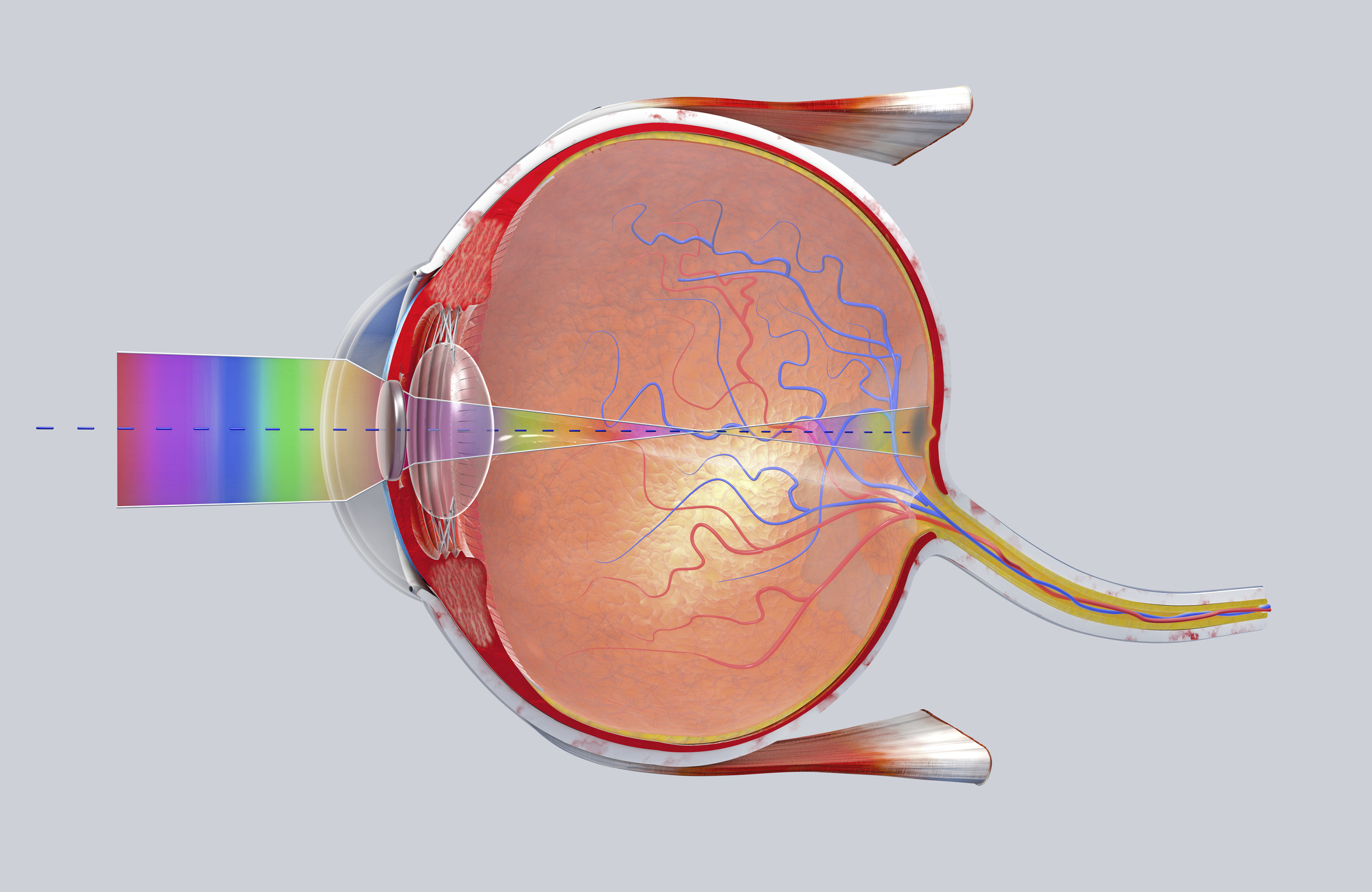
What is it? Scientists successfully used a gene therapy injection to treat patients with Leber hereditary optic neuropathy, or LHON, a rapid-onset disease that leads to vision loss. But there’s more. Surprisingly, administering the treatment in one eye “significantly improves vision in both eyes,” the University of Cambridge reported.
Why does it matter? LHON affects 1 in 30,000 people, primarily men, and usually shows up in patients in their 20s and 30s; within weeks of onset, the optic nerve degenerates such that patients are considered legally blind, with recovery occurring in less than 20% of cases. “As someone who treats these young patients, I get very frustrated about the lack of effective therapies,” said José-Alain Sahel, a professor of ophthalmology at the University of Pittsburgh and senior investigator on a new study published in Science Translational Medicine. “Our study provides a big hope for treating this blinding disease in young adults.”
How does it work? The majority of patients who develop LHON carry a particular genetic mutation on the NT-MD4 gene. The treatment developed by Sahel and colleagues, called mitochondrial targeting, involves the injection of “a viral vector containing modified cDNA” into the vitreous cavity in the back of the eye. In a phase 3 clinical trial, it was administered in one eye to 37 patients who’d suffered vision loss for six to 12 months. The other eye received a “sham injection,” but vision improved in both eyes for more than three-quarters of patients. The researchers say the technology could have potential for treating other mitochondrial diseases.

What is it? Toyota introduced a solid-state car battery that could be “a game changer not just for electrical vehicles but for an entire industry,” according to Nikkei Asia.
Why does it matter? In the electric-vehicle world, the race is on for batteries that can recharge quickly and last for long distances — and are also affordable and compact. According to Nikkei, the electric vehicles now being developed by Toyota will have twice the range of vehicles traveling under similar conditions powered by Li-ion batteries, while not sacrificing interior space.
How does it work? Solid-state batteries use solid electrodes and a solid electrolyte, rather than the “aqueous electrolyte solutions” employed by Li-ion batteries. Toyota’s planned electric vehicle would be able to recharge in 10 minutes; the company it set to unveil a prototype next year.

What is it? Researchers at MIT developed a way to use machine learning and computer simulations to develop better metal alloys.
Why does it matter? As MIT News notes, advanced metal alloys are critical ingredients in everything from “cars to satellites, from construction materials to electronics,” but there are challenges to developing new alloys for specific uses: researchers’ “fuzzy understanding of what happens at the boundaries between the tiny crystalline grains that make up most metals.” The properties of the new materials are determined largely by the behavior of the atoms in the metals that were mixed together to create them, but there’s been no systematic way to predict what those properties will be.
How does it work? The machine learning element comes from taking existing representative cases and getting software to extrapolate “and provide predicted values for a whole range of possible alloy variations.” Among other things, the researchers were able to determine that some alloy combinations that had previously been ruled out as unfeasible were feasible indeed. A database that compiles the findings of this study, published in Nature Communications, has been made publicly available and could help anyone working on developing new metal alloys.
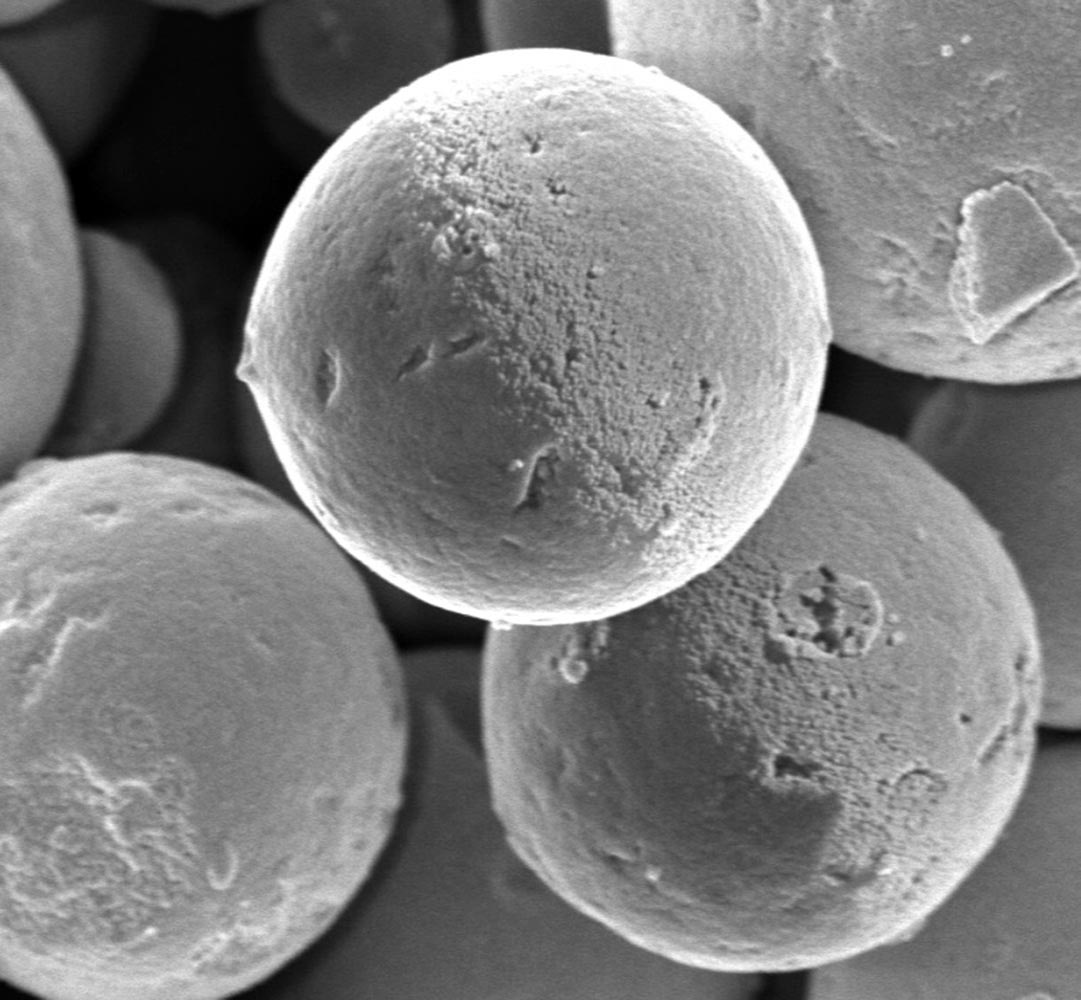
What is it? Researchers at the Dresden University of Technology are investigating synthetic microparticles that exhibit phototaxis — that is, their movement can be influenced by light.
Why does it matter? “We could make use of this property for the targeted control of microrobots,” said TU Dresden’s Juliane Simmchen. “Applications are conceivable in which the particles filter and remove pollutants from liquids or transport medicine through the body, and perhaps even transport information.”
How does it work? Simmchen and colleagues were working with microparticles that — because they include titanium oxide, which reacts with light — are photocatalytic. They noticed that when the particles moved outside the illuminated zone of a microscope, they turned around on their own steam and swam toward the light, a behavior previously known only in microorganisms. In the light, “their swimming direction is stabilized by a combination of physicochemical effects,” according to a press release. “As soon as the particles are no longer exposed to light, there is no energy conversion and the direction of movement is no longer stable.” The authors explained their findings in a paper in Soft Matter: “This interplay shows beautifully how simple physical effects can combine into complex behaviors.”
What Am I, Genetically Engineered Liver?

What is it? Researchers at the University of Pittsburgh School of Medicine used synthetic biology and machine learning to speed up the growth of human livers in the lab.
Why does it matter? According to a news release, the findings — published in Cell Systems — show that “it’s possible to trigger and speed up the maturation of a lab-grown organ without sacrificing precision or control.” That speed is important because, while the human body takes nine months to gestate a liver — which continues to develop after birth — ailing patients in need of a transplant may not have that long to wait. The scientists hope the lab-grown organs might be used in transplants as well as for drug discovery and disease modeling.
How does it work? The team used genetic engineering techniques including CRISPR to “turn a mass of immature liver tissue,” which had been derived from human stem cells, into what they call “designer liver organoids.” Stem cells can be harvested from a patient’s own body, reducing the danger that the body will ultimately reject an organ grown from them. The team transplanted their lab-grown organs into mice with damaged livers, finding that the organs continued to work and that the animals’ lives were prolonged.





