Biologists had long believed that all life evolved from just two types of organisms differentiated by their cells: eukaryotes, creatures like plants and animals whose cells contain a nucleus and membrane-enclosed mitochondria, and bacteria, which have neither mitochondria nor a membrane surrounding their genetic material. But in 1977, American microbiologist Carl Woese discovered that one subset of heat- and salt-tolerant bacteria was actually a “third domain” of life. He called this group archaea.
Scientists now estimate that archaea, which flourish in temperatures approaching 180 degrees Fahrenheit, make up to 20 percent of the Earth’s biomass. But they are only beginning to reveal their secrets.
Researchers at the University of Cambridge and the University of Technology Sydney’s ithree institute have recently published a paper in the journal Nature, illuminating some of evolution’s early steps 2.5 billion years ago. “Archaea and bacteria joined forces early in evolution, resulting in all other complex life we see around us today,” says Iain Duggin, a researcher at the ithree institute.

Top image: Hot springs like Yellowstone’s Grand Prismatic Spring have colonies of heat-loving archaea bacteria living in their bowels. Image credit: Shutterstock Above: Dr. Iain Duggin
Dr. Duggin and his team studied a strain of salt-tolerant archaea from the Dead Sea. “Contrary to its name, the place is actually teeming with life,” he laughs.
They were looking for similarities between proteins produced by eukaryotes and archaea. “We were retracing steps taken by evolution,” Dr. Duggin says. “We wanted to know why the function of certain proteins was conserved.”
The team identified and then deleted individual genes one by one, and observed what happened. “It’s reverse genetics,” Duggin says.
They soon noticed that some genes affected the microbe’s ability to control its shape by changing from a disc to a tube. But Dr. Duggin wanted to dig deeper and document the physical changes taking place inside the microbe.
His archaea were tiny, no more than 2 microns across, 20 times smaller than the width of a human hair. Their innards are basically invisible. The team attacked the problem with the Delta Vision OMX super-resolution microscope from GE Healthcare Life Sciences. The device can observe living organisms in 3D even beyond Ernst Abbe’s diffraction barrier, which for a long time stood as the final frontier for microscopic resolution. “The microscope allowed us to see inside the walls,” Dr. Duggin says. “We were able to resolve details we couldn’t see before.”
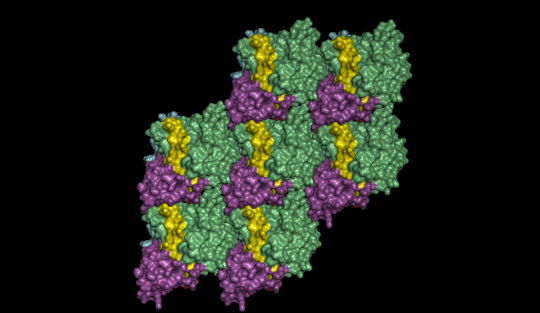
CetZ molecules stick together in a regular pattern to form sheets inside cells. This appears to provide a scaffolding to control cell shape. Remarkably, the overall structure of this sheet is the same in archaeal and human tubulin proteins. Image credit: Dr. Iain Duggin
The team studied the family of proteins, called CetZ, and found that they act like a miniature skeletal system for the archaea cells to control their shape and movement. This “cytoskeleton” allows the cells to transform themselves from a plate shape into a torpedo-like structure for faster swimming. The research suggests that this feature did not evolve with more complex organisms, but may have been inherited from archaea.
This is not just some idle journey into the past. The team wrote that CetZ is “related to a protein in humans that is the target of several major cancer treatments and, in bacteria, the related protein is crucial for cell division and multiplication.”
The human protein is called tubulin and the bacterial version’s name is FtsZ. “While tubulin is a key target in cancer drug development, we believe that FtsZ could be an important target for the development of new antibiotics, potentially enabling the design of anti-infective drugs that inhibit bacterial cell division and growth, with fewer side effects,” Dr. Duggin says.
The archaeal proteins could also illuminate an even older protein ancestor of the tubulin-FtsZ “superfamily” common to the microbial ancestor of all life, including archaea, bacteria and eukaryotes.
Archaea are most likely one of the oldest life forms on Earth. The organisms can survive in extreme cold, heat and salinity, and exist in the soil, sewage, oceans and even oil wells. They make up an estimated 10 percent of the microbial population found within the human gut and are also responsible for biological methane produced by cattle, a major greenhouse gas.
Professor Ian Charles, director of the ithree institute, said in a news release that it was crucial to better understand the function of archaea in nature, and to potentially exploit their properties for industrial and medical applications. “A new type of potentially useful antibiotic called Archaeocin has recently been described that is derived from the archaea,” Charles said. "Archaea provide an untapped source of novel compounds at a time when alternatives are urgently needed given the rapid rise of resistance to existing antibiotics.“





