Mechanical ventilators, machines that keep people breathing when their lungs are seriously compromised, are all over the news lately. They’re an important tool for doctors and nurses working to help the sickest patients suffering from COVID-19, the disease caused by the novel coronavirus. And right now, hospitals around the world need more of them.
Manufacturers, including GE Healthcare, have been stepping up production of ICU ventilators. But in the meantime, doctors are turning to other potentially available options that may be used during this public health emergency — anesthesia machines are one of the tools that may be considered by clinicians.
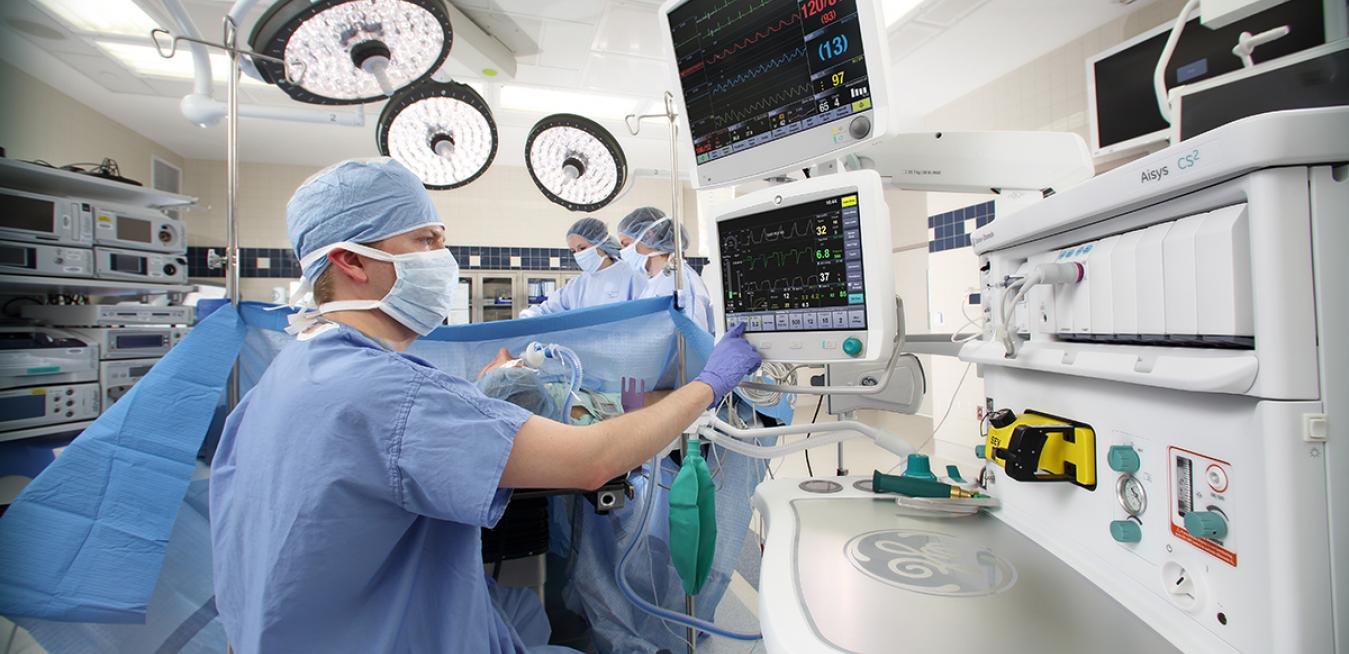
A critical part of the operating room, the anesthesia machine helps keep patients unconscious and breathing during surgery. In alignment with recently issued guidance from the U.S. Food & Drug Administration (FDA), GE Healthcare has provided information to customers on the use of anesthesia machines for patients needing mechanical ventilation during the COVID-19 pandemic.
“Clinicians are sharing that mechanical ventilation, when done correctly and at the right time, can significantly help the mortality rate of the pandemic,” says Lisa Nolan, a certified respiratory therapist and global clinical insights and education leader at GE Healthcare.
Anesthesia machines are indicated for use during surgery, and to provide ventilatory support for a matter of hours not days. The use of these systems to ventilate patients for days or weeks was not validated or cleared by the FDA and is considered off-label use. Clinicians should use traditional ventilators whenever possible. That’s why GE Healthcare, in communication with the FDA and regulatory agencies worldwide, developed information that can be shared with clinicians regarding the off-label use of anesthesia machines for intensive care unit (ICU) ventilation. The FDA also shared: “Wherever possible, health care facilities should use cleared [or approved] conventional/standard full-featured ventilators when necessary to support patients with respiratory failure.”
More than 100,000 GE Healthcare anesthesia machines are currently installed in healthcare facilities worldwide.
 A critical part of the operating room, the anesthesia machine helps keep patients unconscious and breathing during surgery. In alignment with recently issued guidance from the U.S. Food & Drug Administration (FDA), GE Healthcare has provided information to customers on the use of anesthesia machines for patients needing mechanical ventilation during the COVID-19 pandemic. Top and above images credit: GE Healthcare.
A critical part of the operating room, the anesthesia machine helps keep patients unconscious and breathing during surgery. In alignment with recently issued guidance from the U.S. Food & Drug Administration (FDA), GE Healthcare has provided information to customers on the use of anesthesia machines for patients needing mechanical ventilation during the COVID-19 pandemic. Top and above images credit: GE Healthcare.
The company has also posted information on its website and is producing a series of training videos and educational materials that will walk clinicians through the basic functions of an anesthesia machine and differences between anesthesia devices and ICU ventilators.
Some hospitals have begun using anesthesia machines as ventilators — others are likely to follow. It’s an atypical use for the machines, but these are atypical times — and the Anesthesia Patient Safety Foundation says that using anesthesia machines for their ventilating capabilities “certainly should be considered as a life-saving intervention.”
For example, ventilation modes and settings are different on anesthesia machines and ventilators, says Nolan. For one thing, ventilators employ a one-way system — passing air and gas into a patient’s lungs only once — whereas patients hooked up to an anesthesia machine rebreathe some of the air and gases delivered by the device.
Clinicians need to know how to translate between the devices’ settings and adjust their treatment when using an anesthesia machine strictly as a ventilator. For example, anesthesia machines, unlike ICU ventilators, are designed to be turned off and on and should be fully tested at least once a day to maintain proper calibration. GE Healthcare recommends continuing this daily checking process, which means a patient would need to be disconnected and manually ventilated during that time.
Doctors can eliminate the anesthesia aspect of the machine by removing the vaporizers that would normally deliver anesthetic gas to the patient. Then there’s the canister that absorbs carbon dioxide from a patient’s exhalations. The canister, which doesn’t exist on a ventilator, needs to be changed regularly. A clinician may also need to increase the flow of gas on an anesthesia machine, compared with the flow on a ventilator, so that the patient isn’t rebreathing the carbon dioxide, Nolan explains.
One report suggests that up to 70% of surgical procedures are elective and can be rescheduled. Nolan estimates that the average medium to large hospital in the U.S. has 20 to 30 operating rooms. That works out to a lot of anesthesia machines that can be freed up for doctors and nurses doing the urgent work of treating COVID-19 patients.
“It is critical for us to work closely with professional societies, provide easy-to-use education and deliver clinical innovations to clinicians so they can help patients in every way possible,” Nolan says.