Scientists removed HIV from the genomes of mice, used gene editing to help the retina restructure itself, and developed an “EpiPen for spinal cord injuries.” These are big times for bold medical discoveries, but it’s not all serious — we’ve also got soft robotic fish (again!) in this week’s 5 Coolest Things.
 An HIV virus. Top and above images credit: Getty Images.
An HIV virus. Top and above images credit: Getty Images.What is it? Using a combination of treatment and gene-editing therapy, a collaboration between researchers at Temple University’s Lewis Katz School of Medicine and the University of Nebraska Medical Center was able to completely remove “replication-competent” HIV from the genomes containing the virus in living animals — in this case, mice.
Why does it matter? The study, published in Nature Communications, points a way toward a possible cure for HIV, the virus that causes AIDS. Currently, people who are infected with HIV manage their condition with antiretroviral therapy, which suppresses replication of the virus in the body but doesn’t eliminate it altogether; accordingly, treatment has to be followed for the length of a patient’s life.
How does it work? Gene editing has been investigated as another way to combat HIV — but, like antiretroviral therapy, it’s also not able to eliminate the virus completely. In this case two approaches were better than one: The team used a form of treatment called long-acting slow-effective release antiretroviral therapy — or LASER ART — to suppress the replication of HIV, then used CRISPR-Cas9 gene editing to remove remaining fragments of viral DNA. Temple professor Kamel Khalili said, “The big message of this work is that it takes both CRISPR-Cas9 and virus suppression through a method such as LASER ART, administered together, to produce a cure for HIV infection. We now have a clear path to move ahead to trials in non-human primates and possibly clinical trials in human patients within the year.”
 The team designed an instrument to analyze the quality of liquids using the photoacoustic effect, or the generation of sound waves after light is absorbed in a material. The MU scientists believe this might be the first use of this technology to analyze such small liquid samples. Image credit: University of Missouri.
The team designed an instrument to analyze the quality of liquids using the photoacoustic effect, or the generation of sound waves after light is absorbed in a material. The MU scientists believe this might be the first use of this technology to analyze such small liquid samples. Image credit: University of Missouri.What is it? At the University of Missouri, a team of scientists has found a way to use sonar — you know, the technology that helps guide submarines — to determine whether water is safe to drink.
Why does it matter? The technology is cheap and quick, said Professor Luis Polo-Parada, a researcher on the project: “If the water isn’t drinkable, then our method will tell you that something is wrong with the water. For instance, if a facility removes salt from sea water in order for water to be safe for drinking, our method can help alert the facility to potential changes such as an issue with the desalination process.” The team is also looking into how its method could be expanded to help manufacturers measure the quality of olive oil or honey, for example, or measure the levels of sugar in soft drinks.
How does it work? The instrument investigates what’s in the water by way of the photoacoustic effect — that’s “the generation of sound waves after light is absorbed in a material,” according to MU. The researchers used a repurposed laser tattoo-removal machine to blast extremely brief pulses of light into a tape-wrapped cable submerged in liquid. The cable converts the light into sound, whose signals are then analyzed. Gary A. Baker, a chemistry professor who worked on the project, compared it to the way that cymbals work: “Sunlight causes the cymbals to heat up and create a constant ringing sound. Here, on a much smaller scale, we create the same effect by sending flashes of laser light at our tiny homemade cymbal, which is the tape, and measure the speed of the sound that is generated.” The team published its findings in Sensors and Actuators B: Chemicals.
Nano Treatment For Spinal Injury
 Some 12,000 patients in the United States suffer from spinal injury every year, with trauma sometimes resulting in paralysis below the site of the injury. Image credit: Getty Images.
Some 12,000 patients in the United States suffer from spinal injury every year, with trauma sometimes resulting in paralysis below the site of the injury. Image credit: Getty Images.What is it? Researchers at the University of Michigan have developed an “EpiPen for spinal cord injuries” — basically, a quick injection of nanoparticles following spinal trauma that might forestall paralysis.
Why does it matter? Some 12,000 patients in the United States suffer from spinal injury every year, with trauma sometimes resulting in paralysis below the site of the injury. According to researchers, that paralysis can result not just from the injury itself but also from the immune system’s overzealous response — it floods the injury site with immune cells that can create inflammation and scar tissue that prevent the regeneration of nerve cells. The Michigan researchers’ injection seeks to head off that reaction, said Lonnie Shea, a biomedical engineering professor: “In this work, we demonstrate that instead of overcoming an immune response, we can co-opt the immune response to work for us to promote the therapeutic response.” Shea and colleagues, who demonstrated their method in mice, published their work in PNAS.
How does it work? The U. of M. researchers designed injectable nanoparticles that redirect immune cells away from the site of the injury, and that — because they’re nonpharmaceutical — don’t come with the sorts of side effects associated with other similar treatments, like steroid injections. According to the university, “With no drugs attached, the nanoparticles reprogram the immune cells with their physical characteristics: a size similar to cell debris and a negative charge that facilitates binding to immune cells. ... With fewer immune cells at the trauma location, there is less inflammation and tissue deterioration. Second, immune cells that do make it to the injury are less inflammatory and more suited to supporting tissues that are trying to grow back together.”
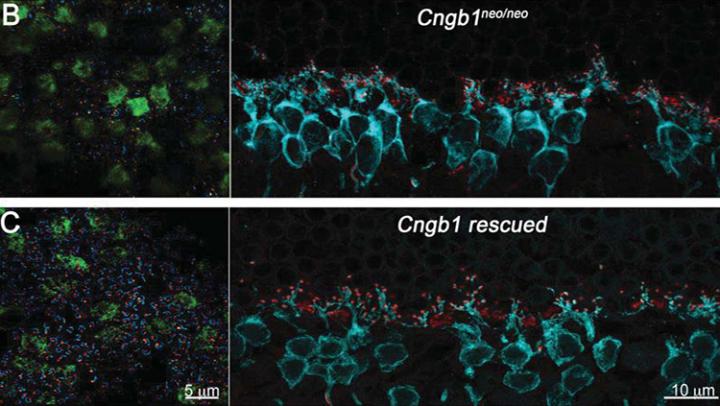 Retinal cells with (bottom) and without gene therapy. Image credit: Wang et al., JNeurosci 2019.
Retinal cells with (bottom) and without gene therapy. Image credit: Wang et al., JNeurosci 2019.What is it? With a little help from gene therapy, the retina can restructure itself, possibly combating blindness, according to new research from scientists at the University of Southern California Keck School of Medicine, the Duke University School of Medicine and the University of California’s Stein Eye Institute.
Why does it matter? “A major cause of human blindness is the death of rod photoreceptors,” the researchers write in a new paper in JNeurosci. Rods are one of the two types of photoreceptors in the eye, the other being cones. According to a release from the Society for Neuroscience, “Current treatments have been developed that can save dying rods, but it was not known if the retina could rebuild itself after treatment, which is a key component of regaining vision.”
How does it work? The team tested their gene-editing hypothesis on mice subjects “with genetically defective rods that mimic developmental blindness disorders in humans.” They found that the rods that received gene therapy were able to reestablish normal light responses and also normal connections to other retinal neurons — findings that, they say, highlight a “surprising degree of plasticity” in the retina itself.
https://www.youtube.com/watch?v=crEHoWgwXX0&feature=youtu.be
What is it? This is a golden age if you’re a researcher working in the soft-robotic-fish space, and now a master’s student at Delft University of Technology in the Netherlands has made another soft robotic leap, claiming to have designed the world’s fastest soft robotic fish. This week he released a video of the creation, which can cruise up to .85 meters per second, or nearly 3 feet.
Why does it matter? According to the video, robotic fins could be a worthy alternative to propellers for underwater craft: Flapping is more efficient, and a lack of “external rotating parts” could allow such craft to operate under the higher-pressure conditions of deeper ocean depths. Plus, it looks cool as heck.
How does it work? The fish uses a single motor that operates a wire — set in a soft, pliant body — to move the tail back and forth, propelling the fish through the water.