Protecting the ticker, a world-first transplant, and hope for aging joints. This week’s coolest things get to the heart of innovating for the greater good.
What is it? Researchers at the Perelman School of Medicine at the University of Pennsylvania used mRNA technology to program immune cells in the body to fight off overactive heart cells that can cause heart failure.
Why does it matter? Fibrosis, a hardening of tissue caused by the overproduction of materials by cells called fibroblasts, can lead to a number of potentially fatal medical conditions, including heart and kidney failure and liver disease. “This technology could turn out to be a scalable and affordable way to address an enormous medical burden,” said Jonathan A. Epstein, chief scientific officer for Penn Medicine and senior author of a new study in Science.
How does it work? The mRNA COVID-19 vaccines work by instructing human cells to produce the coronavirus’s telltale spike protein so the immune system can learn to recognize and attack it. Epstein and his team used the same method to program T cells, which are powerful immune cells, to go after cardiac fibroblasts in mouse models of heart failure. They found that the reprogrammed T cells significantly reduced fibrosis. The effect was temporary, lasting no more than a week. The team hopes to eventually test the technology in human clinical trials.
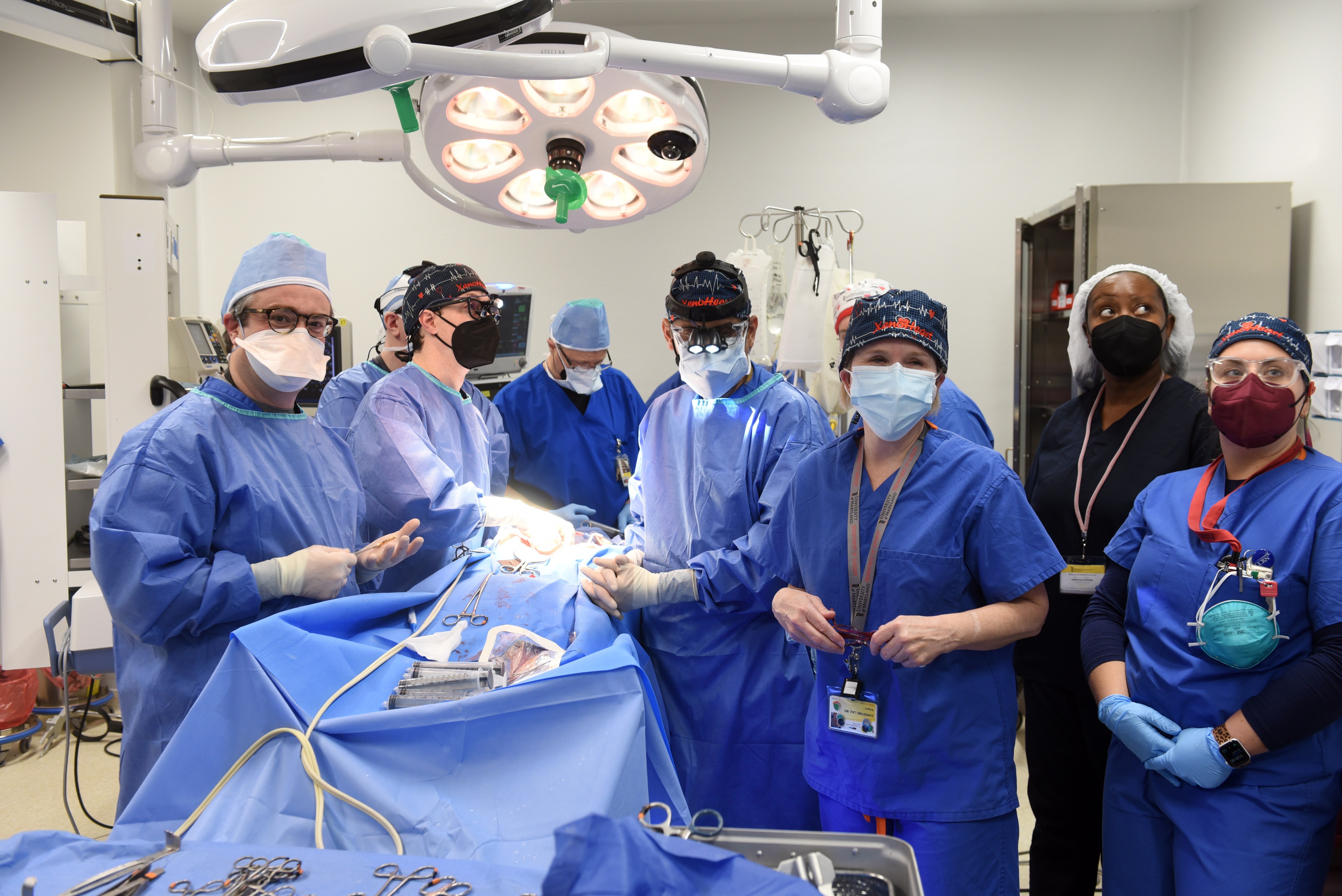
What is it? Surgeons at the University of Maryland Medicine performed the first successful transplant of a genetically modified animal heart into a human.
Why does it matter? More than 106,000 Americans need organ transplants; 17 people die each day waiting for one, according to the Health Resources and Services Administration. Doctors could use animal organs to save potentially thousands of lives but have yet to overcome the problem of organ rejection.
How does it work? The heart was from a pig that had been genetically modified by removing genes that induce human tissue rejection and adding human genes for immunological acceptance. Three days after the transplant, the team reported that the 57-year-old man, who had previously been on life support, was doing well. “We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” said Bartley P. Griffith, who performed the surgery.
Though kelp does not naturally grow in the open ocean, Marine BioEnergy was able to adapt the plant using scaffolding that moves it to the seafloor at night for nutrients and back to the surface during daylight hours. Video credit: Marine BioEnergy.
What is it? California company Marine BioEnergy demonstrated the ability to grow kelp in the open ocean, a proof of concept for raising and harvesting the fast-growing algae for conversion into biofuel.
Why does it matter? Biofuels could help the U.S. transition away from fossil fuels, but they are difficult and expensive to produce at scale. Mass production of kelp in the open ocean could provide a reliable fuel source.
How does it work? Marine BioEnergy’s founders see vast swaths of open ocean as an alternative to growing plants that require lots of land, fresh water and fertilizer. Kelp doesn’t naturally grow in the open ocean. Its habitat is in shallower waters, as it needs to be anchored to the seabed, where it gets vital nutrients, while also being close enough to the surface to get sunlight. The company designed a seaborne scaffolding for growing kelp that is carried to different depths by autonomous submersible drones. The subs bring the algae to the ocean bottom at night so they can feed, then return them to the surface for daytime photosynthesis.
What is it? Bioengineers at the University of Connecticut developed a way to regrow cartilage in arthritic joints.
Why does it matter? More than 30 million Americans have osteoarthritis, a painful condition arising from the breakdown of shock-absorbing cartilage between bones, most often in the fingers, knees and hips. Past attempts to grow back lost cartilage have failed. “The regrown cartilage doesn’t behave like native cartilage. It breaks under the normal stresses of the joint,” said Thanh Nguyen, an assistant professor of mechanical engineering.
How does it work? Nguyen’s lab found that electrical signals are involved in normal cartilage growth, so they created a scaffolding out of a biodegradable polymer nanomaterial that produces piezoelectricity, or electric currents that result from pressure. When the material is implanted in a joint, the activity of walking or running creates small bursts of electricity, which stimulate new cartilage growth without the use of additional growth factors or donor material. When the team inserted the scaffold into the injured knee of a rabbit, cartilage grew back normally. The results were published in Science Translational Medicine.

What is it? University of Minnesota researchers 3D-printed a functional and flexible OLED screen.
Why does it matter? Today, organic light-emitting diode (OLED) displays used in TV and smartphone screens can be produced only in microfabrication facilities. The new 3D-printing method, described in Science Advances, could open OLED innovation to anyone with a capable tabletop printer. “Fully 3D printed OLEDs can potentially be used for important applications in soft electronics and wearable devices,” said lead author Ruitao Su.
How does it work? The Minnesota team used a hybrid approach to print the 64-pixel,1.5-inch-square display with their souped-up 3D printer. They used a standard extrusion printing method for four of the display’s six layers. But for the two layers that emit light — which typically need to be produced using thermal evaporation to achieve adequate uniformity — they switched their machine to spray printing. Even after being bent 2,000 times, the flexible screen continued displaying its scrolling message, “HELLO,” followed by a smiley face.