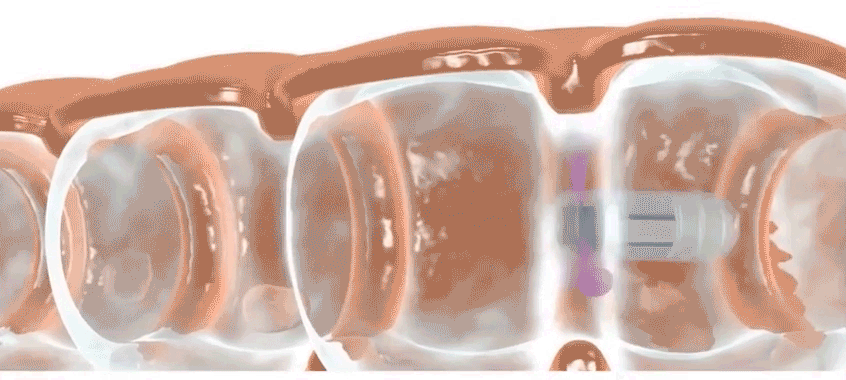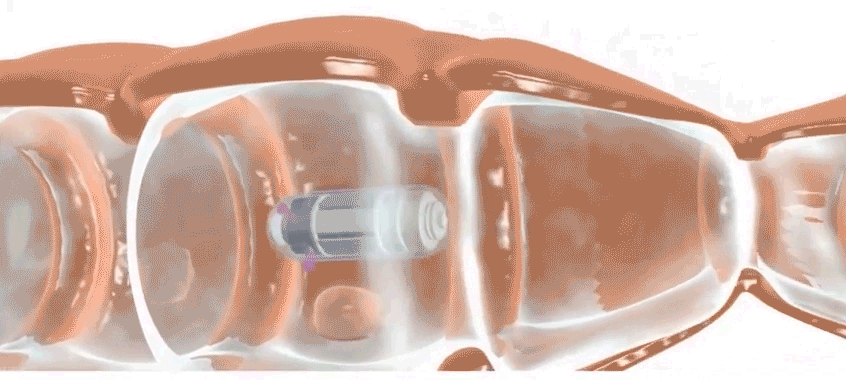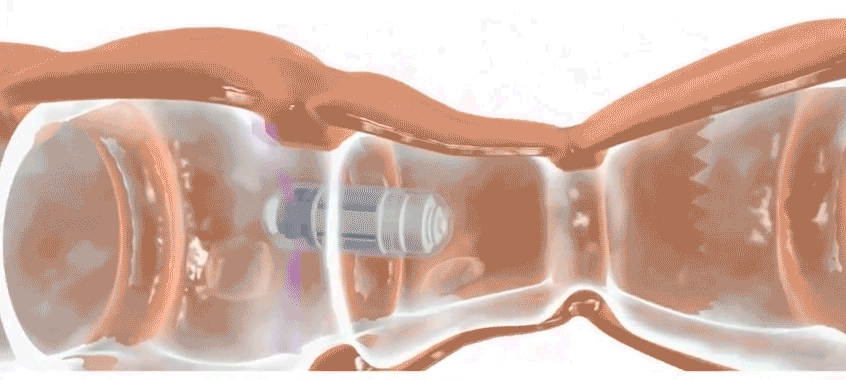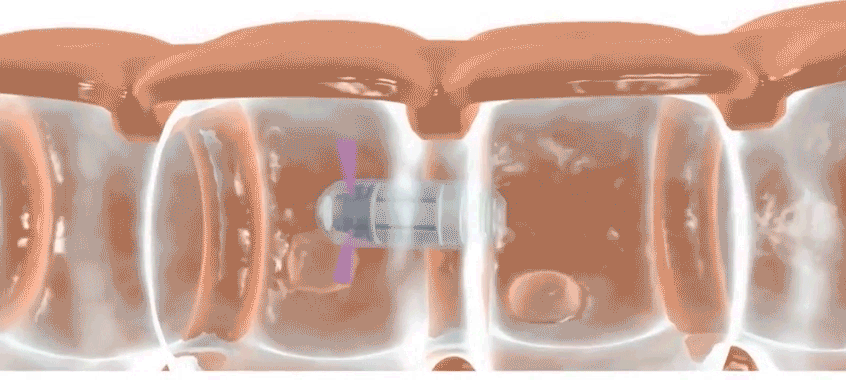
Check-Cap is developing an ingestible scanner that achieves the same purpose. Users simply swallow the device, which is roughly the size of a large vitamin, and then the device spends the next two to three days traveling through the digestive tract, scanning the inside of the colon with precisely timed X-rays. It wirelessly transmits information to a data recorder that users attach to their backs. Doctors then collect the data recorder and analyze the data. Patients should stick to their usual routine during the scan, says Densel. “We want people eating normally, we want them exercising, if that’s what they do,” he says. The capsule doesn’t need to be retrieved after it’s passed through the body.
 “The movie ‘Fantastic Voyage’ is a very applicable analogy to this,” Densel says. Image and GIF credits: Check-Cap
“The movie ‘Fantastic Voyage’ is a very applicable analogy to this,” Densel says. Image and GIF credits: Check-CapCheck-Cap has conducted two “clinical feasibility studies” that enrolled 175 patients in Germany and Israel. “The goal was to assess safety and clinical proof of concept,” Densel says. The company is currently conducting a study at several medical centers in Israel to evaluate safety and clinical performance. Check-Cap is now working with GE Healthcare to boost production capacity of X-ray sources for the capsule.
Check-Cap, which is based near Haifa, Israel, was founded by Dr. Yoav Kimchy, an Israeli physician who recognized that people at high risk for colorectal cancer were resistant to screening. He got the idea for the capsule during his compulsory military service, when he served in the Israeli navy and became familiar with submarine navigation. “Submarines can ‘see’ in dark, murky places by using sonar,” Densel says. “The capsule is basically in the same kind of environment." Kimchy created a technology that uses X-rays instead of sound to map the wall of the colon. “The movie ‘Fantastic Voyage’ is a very applicable analogy to this,” Densel says.
There is a market for the device. The U.S. Centers for Disease Control and Prevention says colonoscopies reduce the number of deaths from colorectal cancer. Still, only 65 percent of Americans between the ages of 50 and 75 are current with screening recommendations. “We wanted to give people another screening option that could improve the patient experience and result in more people being screened,” Densel says.
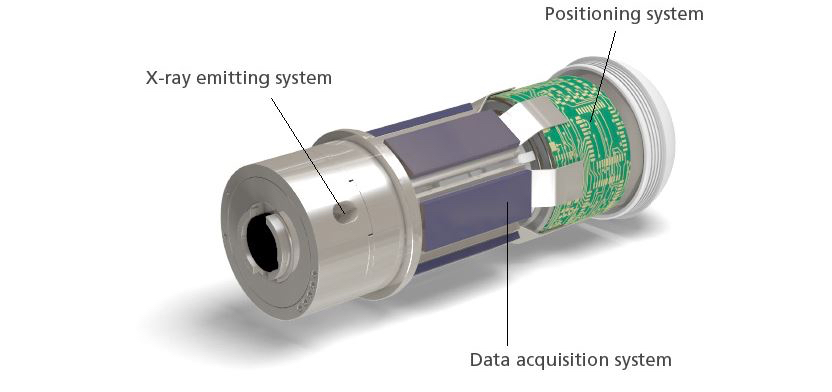
After demonstrating proof on concept, Kimchy and his team settled on a plastic pod that is 3 centimeters long and 11 millimeters wide. It packs an X-ray source, photon detectors, an accelerometer, a special microchip, a radio transmitter, a battery and other parts.
There is no need for emetics before the scan. However, patients must take a tablespoon of a contrast media imaging solution, such as one made by GE Healthcare called Omnipaque, three times a day. “If you did a standard colonoscopy prep and emptied your intestines, the capsule wouldn’t function properly,” Densel says. “The Omnipaque mixes with the content of the bowels and creates a boundary with the colon wall that can be measured.”
[embed width="800"]https://www.youtube.com/watch?v=pjBj7IIuPWg&feature=youtu.be[/embed]
The capsule’s position and movements are continuously tracked as it travels through the body. It scans only when it is in the colon and in motion, which prevents re-scanning of the same area and minimizes X-ray exposure.
Next, a radio transmitter sends the information to a thin data recorder attached to the patient’s back. “We can triangulate where in the colon the capsule is and make a map of the wall,” Densel says. “When we find a feature that looks like a polyp, we know where it is.”
When the capsule exits the body, the patient returns the trackers with the data to the doctor for analysis. Densel says the capsule is designed to utilize an X-ray source with a short half-life so that it doesn’t have to be retrieved at the end of its journey.
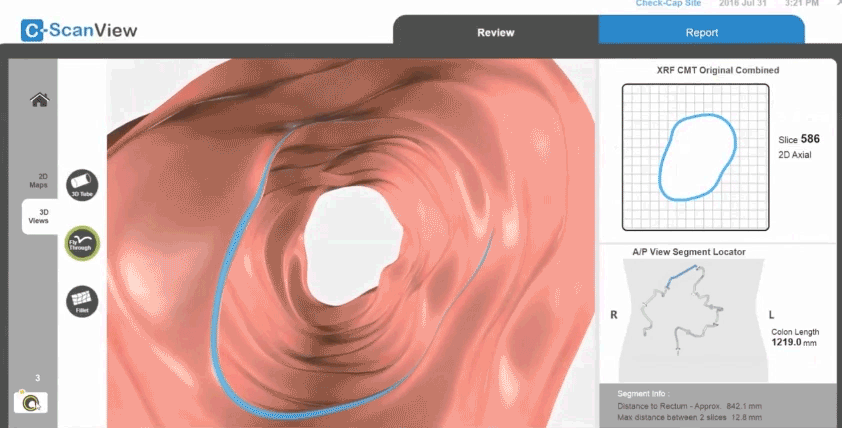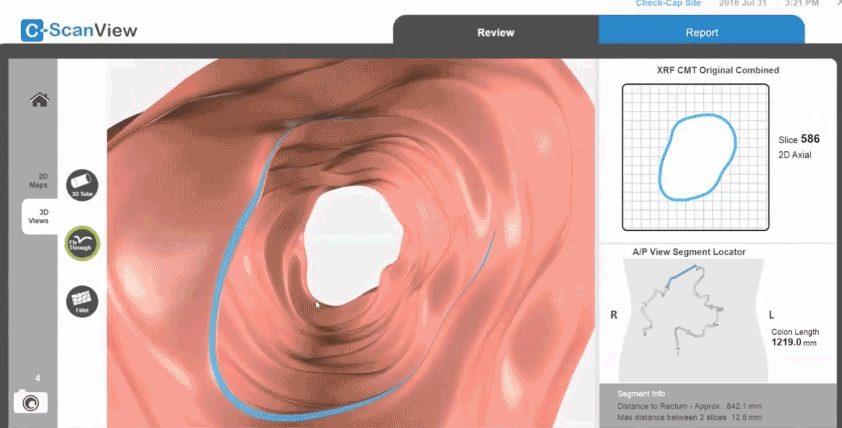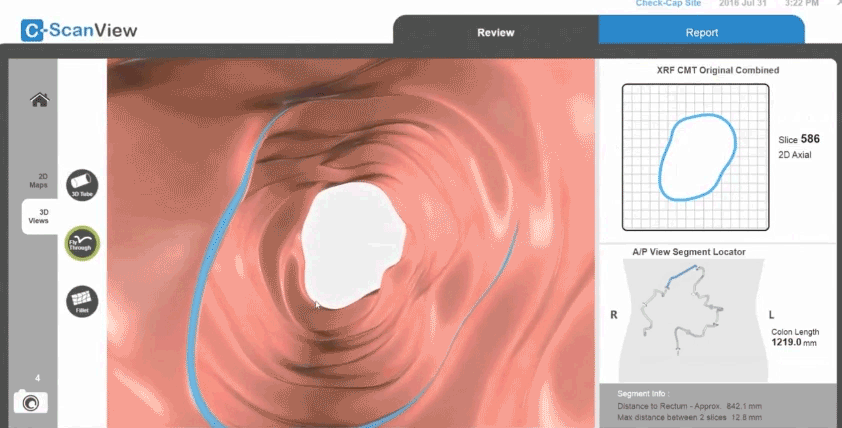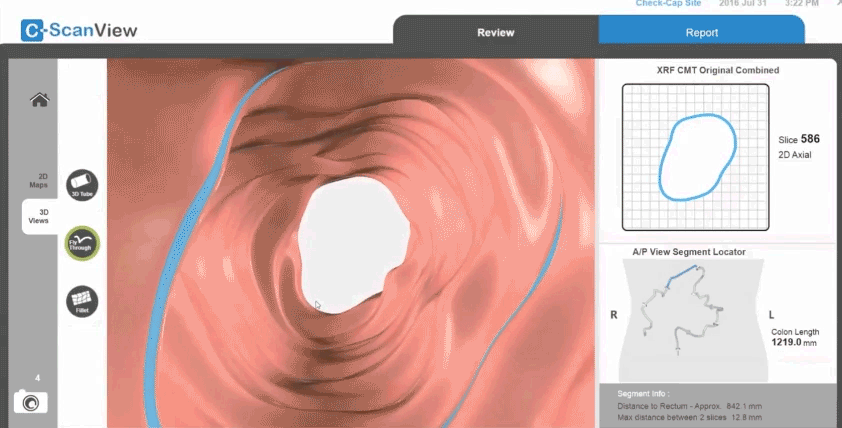
Check-Cap, which has been around since 2005, is now planning clinical studies in the U.S. and Europe to measure the technology’s effectiveness. “We are going to see how well the capsule did compared to the fecal immunochemistry test, which is common in Europe, and colonoscopy, which accounts for 90 percent of screening in the U.S.,” Densel says.
The company is collaborating with GE Healthcare, which manufactures radiation sources for the drug and medical device industry, to create a set of manufacturing and distribution processes that will allow Check-Cap to boost production and serve global markets.